All Features

Ryan E. Day
BioBridge Global (BBG) is a parent organization for four subsidiary organizations, three of which are involved in production activities, and they’re all around regenerative medicine, including blood components, clinical laboratory testing, and cell and tissue therapies. Organizations in the life…

Taran March @ Quality Digest
These days, even regulatory agencies must innovate if they expect to keep pace with the speed of doing business. The U.S. Food and Drug Administration is no exception, and this year especially it has challenged itself to find ways to enhance efficiency and update old regulations. Quality Digest has…

Dirk Dusharme @ Quality Digest
The Dec. 31, 2018 deadline looms for medical device companies that sell their devices in Canada. On that day, any company that sells medical devices to Canada will either need to hold an MDSAP certificate or show proof that they are on track to be MDSAP certified, or they won’t be able to sell…

Mike Richman
The future is the ultimate abstraction; anyone who has ever attempted to discern the nature of tomorrow by looking at the yesterdays leading up to today knows that prediction is a fool’s errand. That’s the unfortunate reality for weather forecasters, stockbrokers, sports bookmakers, political…

Ryan E. Day
One of the unique aspects of Finch Therapeutics is that although its product does not fall easily into any regulated category and thus is not FDA-approved, the company has been working closely with the agency for at least five years. The FDA has broad jurisdiction to regulate all health products,…

Matthew M. Lowe
Life science companies play a major role in the global economy, with revenues expected to reach a staggering $1.5 trillion by 2020.1 Such a rosy forecast is likely to attract innovators and encourage current industry players to blaze new trails. Whether new or established, life science companies…

Andreas Engelhardt
An international standard that specifies requirements for an occupational health and safety (OH&S) management system, ISO 45001:2018—“Occupational health and safety management systems–requirements” replaces OHSAS 18001 as the primary OH&S standard used internationally. It follows other…

Grant Ramaley
The Dental Trade Alliance learned from its members in February 2018 that the Canadian Health Ministry (“Health Canada”) had contacted the Standards Council of Canada (SCC) and the British Standards Institution (BSI). Health Canada had ordered these certification bodies to stop issuing ISO 13485…

Richard Pazdur
During the past decade, advances in understanding of cancer biology have led to the development of targeted treatments that are more effective than the chemotherapies of the past century. These therapies are demonstrating response rates large in magnitude or response durations prolonged in early…

Mike Richman
‘Culture” is one of those business-speak words that’s used a lot, but for a good reason—having the right one is the key to unlocking your company’s quality potential. On the other hand, nothing will overcome a poor culture. Do you know which you have? We explored these issues during the Aug. 10,…
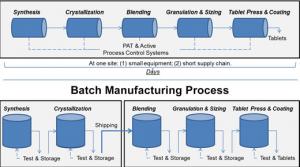
Scott Gottlieb
There’s new technology that can improve drug quality, address shortages of medicines, lower drug costs, and bring pharmaceutical manufacturing back to the United States. At the U.S. Food and Drug Administration (FDA), we’re focused on propelling these innovations, collectively referred to as…

Vanessa Burrows, Suzanne Junod, John Swann
During the early 20th century, Americans were inundated with ineffective and dangerous drugs, as well as adulterated and deceptively packaged foods.
A cosmetic eyelash and eyebrow dye called Lash Lure, for example, which promised women that it would help them “radiate personality,” in fact…

Matthew M. Lowe
The medical marijuana industry is being heralded as the new frontier in the life sciences, thanks to the potential of cannabis-derived products in treating ailments that range from chemotherapy-induced nausea to epilepsy and neuropathic pain. If you’re a startup in the industry, what does this mean…

Janet Woodcock
The staff of the U.S. Food and Drug Administration’s (FDA) Center for Drug Evaluation and Research (CDER) always tries to utilize cutting-edge science and up-to-date process management, befitting our stature as the global “gold standard” in drug regulation. Maintaining that standard requires us to…

Jon Speer
“I wish there was a way for the FDA to give me a heads-up about my stuff, prior to submission….”
That sentiment was really the basis behind the U.S. Food and Drug Administration’s (FDA) presubmission tool, as I was discussing recently with medical-device quality assurance and regulatory affiars…

Malvina Eydelman
The U.S. Food and Drug Administration’s (FDA) Breakthrough Devices Program is beginning to show important results for patients since it was established in late 2016 under the 21st Century Cures Act to help patients gain timely access to breakthrough technologies.
Consider Second Sight Medical…

AssurX
Recent FDA warning letters indicate that many drug manufacturers do not have their manufacturing in a state of current good manufacturing practices (CGMPs) control. During the first half of 2017, the FDA cited adulterated products and insanitary conditions as the two most common violations in drug…

Scott Gottlieb, Jeffrey Shuren
In recent days, the U.S. Food and Drug Administration (FDA) has committed to several new policies that will modernize the agency’s approach to regulation in the medical device system.
For instance, we announced our intention to propose an alternate approach to the traditional 510(k) clearance…

Jon Speer
Complaint handling continues to be one of the biggest reasons medical device companies receive 438s and warning letters from the U.S. Food and Drug Administration (FDA). Companies have a lot going on once a medical device has reached the market, and it can be challenging to keep up with…

Scott Gottlieb
Twice a year the federal government publishes the Unified Agenda of Federal Regulatory and Deregulatory Actions (Unified Agenda), which provides the American public with insight into regulations under development or review throughout the federal government. For the U.S. Food and Drug…

Lou Valdez, Dara Corrigan, Peter Stein
Regulatory experts from around the world, including the Food and Drug Administration (FDA), gathered recently to discuss issues such as regenerative medical products, international collaboration to fight antimicrobial resistance (AMR), and developing strategies to combat substandard or falsified…
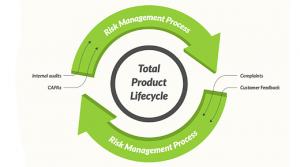
Jon Speer
What exactly is a risk-based quality management system (QMS)? This is a timely topic to get into. In 2016, ISO 13485—“Medical devices”—“Quality management systems” was updated, and one of the key concepts presented is the idea of a risk-based QMS.
Historically, regulations have almost exclusively…

Grant Ramaley
I have written previously about the Medical Device Single Audit Program (MDSAP) created by the International Medical Device Regulators Forum (IMDRF). MDSAP is viewed as a single audit covering the United States, Canada, Brazil, Australia, and Japan. The intent was to establish one medical-device…

Mike Richman
QDL co-host Dirk Dusharme was on vacation for our Nov. 10, 2017, episode, but we ably covered for his absence with some thought-provoking stories and great guests. Let’s take a look:
“What Really Causes Workplace Stress” A multidisciplinary team of researchers at the University of Southern…

Brandon McFadden
The food labeling craze coupled with banner headlines about the dangers of gluten, genetically modified organisms (GMOs), and hormones are leading to increasingly absurd results.
For example, you can now buy “premium” water that’s not only free of GMOs and gluten but certified kosher and organic…